 Make separating gel as following formula in a suitable beaker.All following operations related with apparatus should follow manufacture’s instructions. Assemble mutil-casting chamber according to the manufacturer’s instructions.Gradient gel former: Bio-Rad Model 485 Gradient Former.Minigel apparatus: Bio-Rad Mini-Protean 3 Dodeca Cell.Coomassie gel destain solution: 30% ethanol, 10% acetic acid.Coomassie gel stain solution: 0.1% Coomassie blue R-250, 30% ethanol, 10% acetic acid.Electrophoresis buffer: 0.25M Tris Base, 2M Glycine, 1% SDS.TEMED(N,N,N’,N’-tetramethylene-ethylenediamine).Acrylamide stock solution: 29% acrylamide plus 1.0% bis-acrylamide.Separating gel buffer: 1.5M Tris-HCl, pH8.8, 0.4% SDS.Stacking gel buffer: 0.5M Tris-HCl, pH6.8, 0.4% SDS.The cell lysate is evaluated as qualified, if the WB image shows five bands. The primary antibody we used is the antibody against the marker proteins. The cell lysate is evaluated as qualified, if the bands are clear and have no obvious smear. 
Quality control a) Test cell lysate by SDS-PAGE.b) Mix the cell lysate with SDS loading buffer to make the desired final concentration.Measure protein concentration using cell lysis-compatible protein assay (BCA protein assay). Dilute the supernatant lysate and the whole lysate without centrifugation at 1:4, 1:8 and 1:16 with 1XPBS. Then collect supernatant into an appropriately labeled tube (for the detection of membrane bound proteins, use the insoluble, cell pellet). Measure concentration of cell samples a) Transfer contents to a microcentrifuge tube.b) Disrupt tissue with ultrasonicator.Preparation of tissue extracts a) Thaw the frozen, raw tissue sample by vortexing and repeat the freeze-thaw cycle twice.Vortex the mixture for 5min and store at -20C. c) Add cell lysis buffer to cell extracts (1ml lysis buffer /5×10 7 cells). When the concentration reach 10 6 cells/ml, detach cells, spin down and wash twice with 1XPBS. b) Incubate cells for about three days and count the cells. Then spin down cells and re-suspend the pellet in DMEM with 10% FBS to make the final concentration around 105 cells/ml, and incubate at 37C, 5%CO2. Ii) Adherent cells: digest adherent cells in Cell Detaching Trypsin Buffer. I) Suspension cells: spin down cells and re-suspend pellet in RPMI 1640 with 10% FBS to make the final concentration around 105 cells/ml, and incubate at 37C, 5%CO2. Cell culture and harvest a) Cell subculture.Cell Lysis Buffer (1% NP40, 0.5% DOC, 1mM EDTA, 65mM Tris-HCl pH=6.4, 1mM PMSF, 1ug/ml Aprotinin, 1ug/ml Leupeptin, 1ug/ml Pepstatin).Cell Detaching Trypsin Buffer: 0.25% trypsin in 0.53M EDTA solution.SDS-Loading Buffer: 0.05M Tris-HCl, pH6.8, 0.1M DTT, 2%SDS, 10% glycerol, 0.1% bromophenol blue.Phosphate Buffered Saline 1X PBS, pH 7.4. 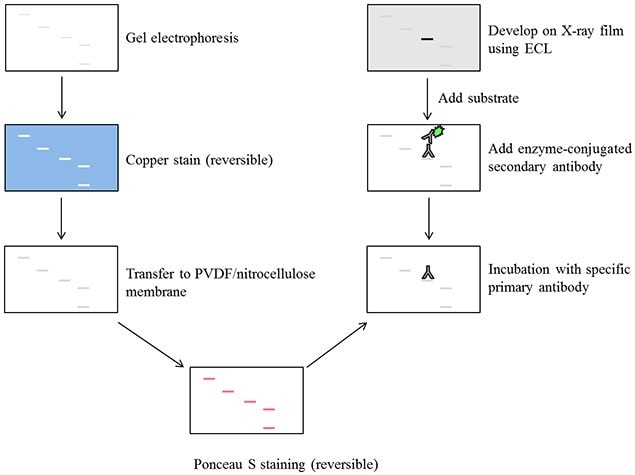
Please consult the product data sheet for the appropriate concentration of primary antibody and any other special conditions. see example western blots from our validation program! Procedure: Enzyme substrate is then applied and the membrane is visualized for the presence of signal. 
The membrane is then probed with an antigen specific antibody which is then itself detected using an enzyme conjugated antibody. Typically, protein samples are resolved by their size by gel electrophoresis and transferred onto a membrane. Western Blotting / Immunoblotting (WB / IB) Description:Ī western blot is a technique used to identify the presence of an antigen in a particular tissue homogenate or protein extract. Western Blotting/Immunoblotting (WB/IB) ProtocolĮnzyme-Linked ImmunoSorbent Assay (ELISA) Protocolīlocking Peptide Competition Protocol (BPCP)
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
